Episodes

Tuesday Mar 26, 2019
Heterogeneous Sodium in Ventricular Myocytes
Tuesday Mar 26, 2019
Tuesday Mar 26, 2019
Is sodium evenly or unevenly distributed in the subsarcolemmal space? Intracellular sodium is an important regulator of cardiac contractility and plays a role in cardiac arrythmias. Therefore, understanding how and why sodium may be differentially distributed in the subsarcolemmal space is critically important. Associate Editor Mario Delmar (New York University) interviews lead author Jonas Skogestad (University of Oslo) and content expert Sand Despa (University of Kentucky) about the novel studies by Skogestad et al. Using both patch-clamping and mathematical modeling, Skogestad and co-authors challenge the current paradigm by suggesting that subsarcolemmal sodium may exist in not just one, but rather many, compartments. The authors found that, after activating the sodium channel for a short amount of time, the sodium released into the subsarcolemmal space did not affect Na+/K+-ATPase (NKA) activity. Did the same hold true for longer activation times, and what does this indicate about the possible interaction between the sodium pump and sodium channels? Listen now.
Jonas Skogestad, Glenn Terje Lines, William E. Louch, Ole M. Sejersted, Ivar Sjaastad, and Jan Magnus Aronsen Evidence for heterogeneous subsarcolemmal Na+ levels in rat ventricular myocytes Am J Physiol Heart Circ Physiol, published January 18, 2019. DOI: 10.1152/ajpheart.00637.2018

Friday Mar 22, 2019
Mitochondrial Oxidative Stress in Smoking and Hypertension
Friday Mar 22, 2019
Friday Mar 22, 2019
Tobacco smoking is still a major global public health risk, despite the well-known facts that smoking increases the risk of cardiovascular disease and hypertension. What is the mechanism by which tobacco smoking induces mitochondrial oxidative stress? Editor-in-Chief Irving H. Zucker (University of Nebraska Medical Center) interviews lead author Sergey Dikalov (Vanderbilt University Medical Center) and content expert Andreas Beyer (Medical College of Wisconsin) about the new study by Dikalov et al that uses a unique chronic tobacco smoking model in both human and animal tissues to determine that smoking induces oxidation of mitochondrial cardiolipins. Dikalov and co-authors performed novel catalase experiments to show that smoking interferes with cell signaling pathways, and that specific scavenging of mitochondrial catalase can prevent alterations of vascular functions. Short-term (just 2 weeks!) of tobacco smoking showed marked reductions in SIRT3, which is a key mitochondrial deacetylase and a marker of longevity. Did smoking cessation return SIRT3 to normal healthy levels? Listen and learn.
Sergey Dikalov, Hana Itani, Bradley Richmond, Aurelia Vergeade, S. M. Jamshedur Rahman, Olivier Boutaud, Timothy Blackwell, Pierre P. Massion, David G. Harrison, and Anna Dikalova Tobacco smoking induces cardiovascular mitochondrial oxidative stress, promotes endothelial dysfunction, and enhances hypertension Am J Physiol Heart Circ Physiol, published February 27, 2019. DOI: 10.1152/ajpheart.00595.2018
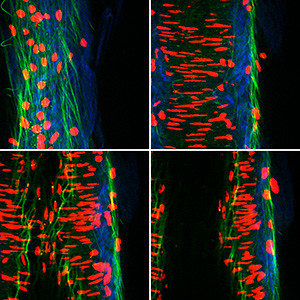
Wednesday Mar 06, 2019
Vasoconstriction in White and Brown Adipose
Wednesday Mar 06, 2019
Wednesday Mar 06, 2019
How is adipose tissue vascular reactivity impacted by obesity? Editor in Chief Irving H. Zucker (University of Nebraska Medical Center) interviews lead author Lisa Lesniewski (University of Utah) and content expert Camilla Wenceslau (University of Toledo) about this central question in the new study by Hazra et al. To explore the differential activity of feed arteries from both white and brown adipose tissue to important vasoconstrictors, Lesniewski and co-authors used a pressure myography system to examine vasoreactivity in excised feed arteries from normal and high-fat diet fed mice. Despite greater non-receptor mediated vasoconstriction in arteries from brown adipose tissue, the authors did not find an increase in receptor mediated vasoconstriction in brown adipose tissue compared to white adipose tissue. In general, Lesniewski and colleagues found that a high-fat diet lowered reactivity to the receptor-mediated agonists. Could stimulating blood flow in the vasculature of brown adipose tissue have clinical implications for fighting the obesity epidemic? Listen and learn more.
Sugata Hazra, Grant D. Henson, R. Colton Bramwell, Anthony J. Donato, and Lisa A. Lesniewski Impact of high-fat diet on vasoconstrictor reactivity of white and brown adipose tissue resistance arteries Am J Physiol Heart Circ Physiol, published February 12, 2019. DOI: 10.1152/ajpheart.00278.2018

Tuesday Mar 05, 2019
HIF1 Regulates miR-29c and SERCA2
Tuesday Mar 05, 2019
Tuesday Mar 05, 2019
How does HIF1 regulate microRNAs in the heart? This is the question we explore in our latest podcast. Listen as Associate Editor Junichi Sadoshima (Rutgers New Jersey Medical School) interviews authors Ralph Shohet and Allison Williams (both of University of Hawaii), along with content expert Asa Gustafsson (University of California-San Diego), about the intriguing new work by Williams et al. It is known that miR-29c is a critical mediator of HIF1 for downregulation of SERCA2, which is commonly seen in heart failure. Using a tetracycline-inducible transgenic mouse model with an oxygen-stable form of HIF, the authors observed deeper levels of HIF action in the heart. What consequences of stable HIF1 activation did the authors uncover? How closely does isolated HIF expression mimic authentic ischemia? Listen and find out.
Allison Lesher Williams, Chad B. Walton, Abigail Avelar, and Ralph Victor Shohet HIF-1 regulation of miR-29c impairs SERCA2 expression and cardiac contractility Am J Physiol Heart Circ Physiol, published February 20, 2019. DOI: 10.1152/ajpheart.00617.2018

